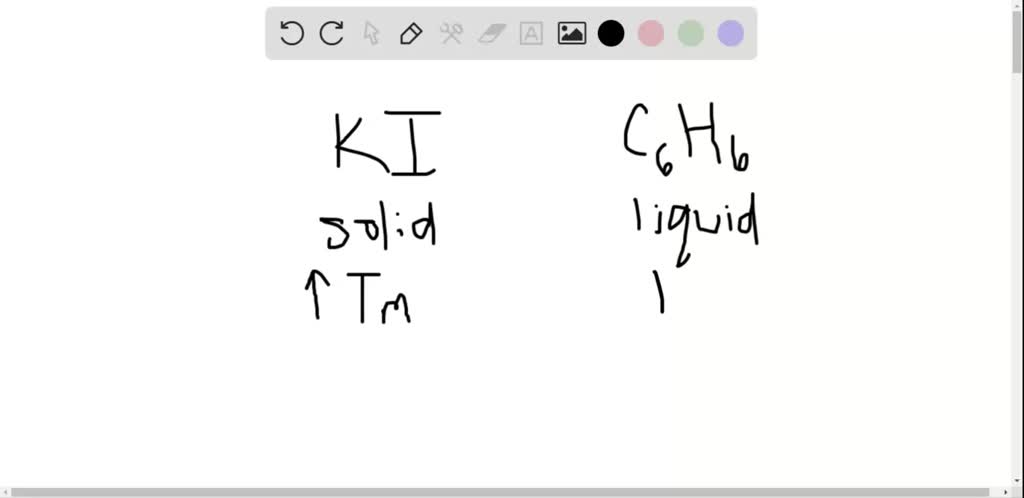 
The number of ions of each element is written as a subscript number after the atomic symbol. Ĭharges of the ions in the formula of the compound are not written. Exampleįinally, construct the formula – two iron ions and three oxide ions are needed. This means there need to be two iron ions and three oxide ions. Since the iron has three positive charges and the oxide has two negative charges, the only way to cancel the charges is to have six of each. As a second example, magnesium chloride has the formula MgCl. This means we have to have the same number of positive charges as negative charges. Its formula is NaCl, which is electrically neutral because sodium ion is +1 and chloride ion is -1. Next, cancel out the charges – the overall charge of an ionic compound must be zero. įirstly, identify the ions – iron(III) oxide contains iron(III) ions (Fe 3+ ) and oxide ions (O 2– ). As hydroxide ions are made of a group of atoms, we must put them in brackets in the formula. Construct the formula – one magnesium ion and two hydroxide ions are needed.In this case, magnesium has two positive charges which means that two hydroxide groups are needed to cancel out the charges completely. You must consider the charge of the ions when writing the formula for an ionic compound from its name, however. 
Cancel out the charges – the overall charge of an ionic compound must be zero.Identify the ions – magnesium hydroxide contains magnesium ions (Mg 2+ ) and hydroxide ions (OH – ).is CH 2 and its relative formula mass is 42. Sometimes an ionic formula contains compound ions – a group of atoms bonded together with an overall charge. The formulae given for ionic compounds, giant molecules and metals. Construct the formula – one of each ion is needed.Since the sodium has one positive charge and chloride has one negative charge, only one of each is needed to cancel the charges. This means the same number of positive charges as negative charges is needed. Identify the ions – sodium chloride contains sodium ions (Na + ) and chloride ions (Cl – ).In Unit 1 you are expected to be able to use the table of common ions to identify the ions present in a compound and then construct the formula of the compound from the ions. The details of how ions are formed are not required for this topic. For example, KCl, an ionic compound that contains K and Cl ions, is named potassium chloride. Reactions between metals and non-metals include: For binary ionic compounds (ionic compounds that contain only two types of elements), the compounds are named by writing the name of the cation first followed by the name of the anion. The resulting compound is called an ionic compound. When metals react with non-metals, electrons are transferred from the metal atoms to the non-metal atoms, forming ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
